
M. ohione, with typically small male (left) and larger female (right) Distribution of M. ohione in the Mississippi River system:
(black line, present day distribution; red dots, pre-1930's)
There are many species of caridean
shrimps that live in fresh water: in lakes, streams, rivers. Many
of these are completely adapted to the f.w. habitat in that they pass the
entire life cycle in fresh water. The planktonic larval stages of
their marine ancestors are reduced are abbreviated or completely eliminated,
i.e., embryos hatch out as benthic juveniles. However, many other
species have not lost their connection to the sea and, although the adults
live all or most of their lives in fresh water, the larvae are planktonic
and require some salinity for development. They must develop in the
brackish water of estuaries or in coastal seas. Afer larval development,
the newly settled benthic juveniles must migrate back up streams and rivers
to the adult habitat. Some of these juvenile migrations have been
observed and studied in recent years and can be quite spectacular.
This life cycle is termed "amphidromous" and is exhibited by many species
in the families Atyidae and Palaemonidae (esp. the genus Macrobrachium).


M. ohione, with
typically small male (left) and larger female (right)
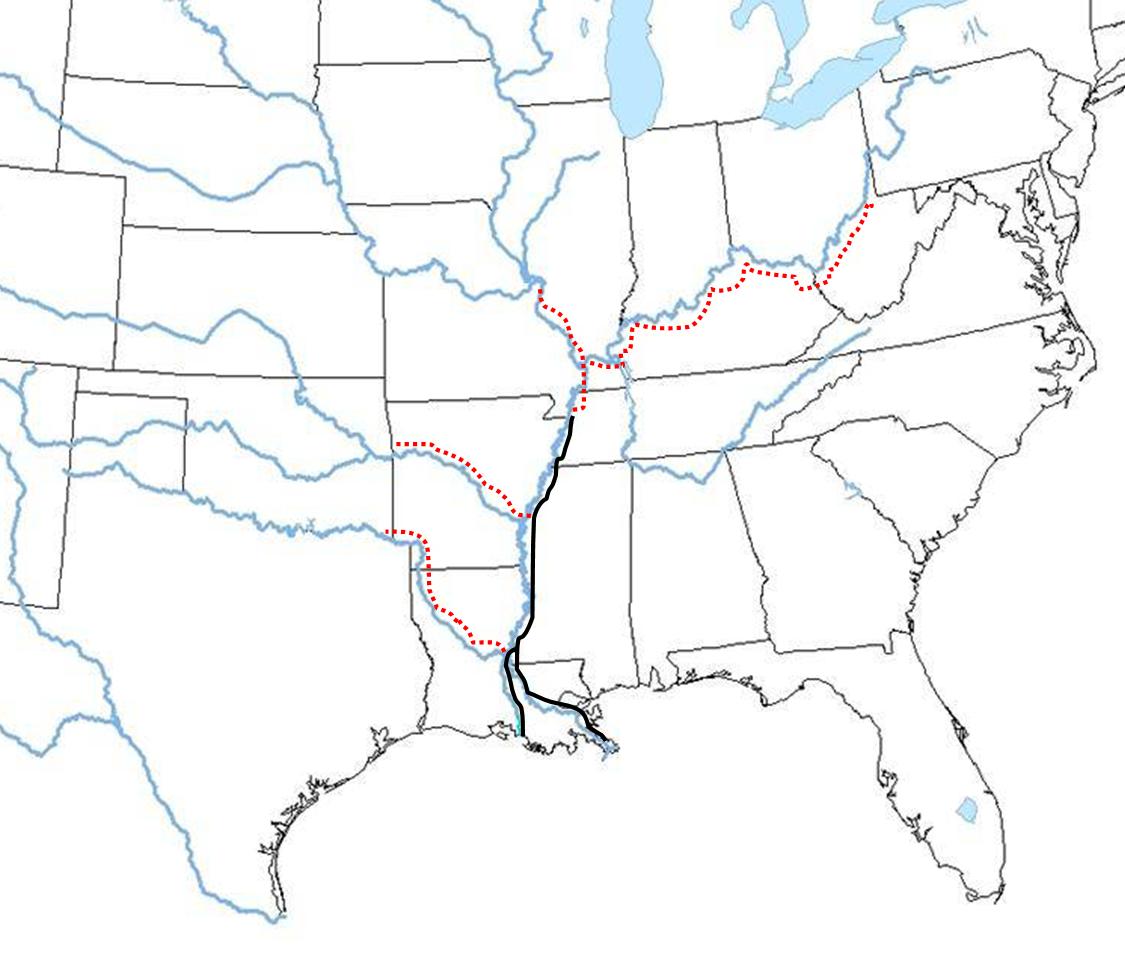
Distribution of M. ohione in the Mississippi River system:
(black line, present day distribution; red dots, pre-1930's)
M. ohione is one of several species of Macrobrachium that inhabitat coastal river systems emptying into the Gulf of Mexico and the s.e. Atlantic coast of the U.S.A. All species were formerly more abundant but human impacts on rivers (especially dams and other river control measures) have considerably reduced populations. Until about the mid-1930's, M. ohione was so abundant in the Mississippi and Ohio river systems that it supported a small but significant fishery for both human consumption and for sale as bait. It is the still the favorite bait item for commercial fishermen in the lowr part of this river system (especially for catfish trotlines). The shrimp must have served as a major prey item for river fishes, and its decline in the northern part of its range most likely has affected fish productivity. It is still fairly abundant in the lower Mississippi and Atchafalaya rivers in Louisiana, where we are studying it.
M. ohione females must either release (hatch) embryos
upstream, with the first stage larvae drifting down to the sea OR the females
may simply make the trip themselves, migrating downstream to estuaries
to hatch the embryos for larva larval development. We are studying
both the female downstream migration as well as the "return" upstream juvenile
migration after larval development. This research is significant
not only for understanding the evolution of amphidromous life cycles but
also to determine how human impacts on these migrations may have caused
the decline of this ecologically and commercially important river species.
If human impacts are responsible, our work may contribute to the conservation
of the species where it is still abudant and to its restoration in northern
parts of its range where it was once so important. Our work is supported
by the NOAA Louisiana Sea Grant College Program,
which we gratefully acknowledge (grant No. NA06OAR4170022, Project
No. R/SA-04 to RTB and Louisiana State University to continue this project
on river shrimps. Statements, findings, conclusions, and recommendations
are those of the authors and do not necessarily reflect the views of Louisiana
Sea Grant or the U.S. Department of Commerce).
Larval Biology
We are studying larval requirements to answer the questions: how long can hatching (stage 1) larvae drift downstream in fresh water before encountering the saltwater necessary for further development? What is the optimal salinity for the critical stage 1 to stage 2 molt? This will help us determine whether females must migrate all the way to the correct salinity in estuaries to hatch larvae or whether they can simply migrate to within a larval "drifting distance" upriver to release the stage 1 larvae.
STAGE 1 to STAGE 2 MOLT IS CRITICAL


Stage 1 larva:
Does NOT feed (relies on yolk droplets left over from the embryo)
Stage 2 larva: Begins
to feed and go to further larval stages
Previous work has shown that Stage 1 larvae maintained in fresh water begin to die after 5 days; those maintained in partial seawater after hatching (15 ppt) are able to molt to stage 2 within a few days and thus begin feeding and continue throught larval development (Bauer and Delahoussaye, 2009).
How long can stage 1 larvae drift in fresh water before arriving in saltwater and what is the optimal salinity needed for the stage 1 to stage 2 molt? The coastal estuaries (e.g., Atchafalaya Bay) of Louisiana have very low salinities during the breeding season.



Females (with embyros) maintained individually
in buckets Hatching bucket
with aeration
Female in hatching bucket



Replicates of different larval treatments in incubator
Culture dishes with larvae and aeration
Very slow aeration (~1 air bubble per second)
 ULL
undergraduate Nick Rome, with funding from the LA Sea Grant Undergraduate
Research Opportunities Program (UROP), performed an experiment, using
a factorial design, to determine the optimal drifting (exposure) time in
fresh water and optimal salinity for the critical stage 1 to stage 2 larval
molt. Hatching larvae are obtained by collecting females from the
field and maintained on laboratory water tables until hatching. Alternately,
females with maturing ovaries are brought in to the lab, maintained with
some of the males. When such females molt, the ovaries are ready
for spawing, mating occurs, and embryos are attached beneath the female
for incubation and later hatching (stage 1 larvae shown swimming at
left). Females near hatching are isolated in hatching tanks and
stage 1 larvae are collected for the experiments. In the experiments,
larvae are maintained in fresh water in treatments which vary in
the number of days in fresh water prior to introduction into seawater
of differing salinities.
ULL
undergraduate Nick Rome, with funding from the LA Sea Grant Undergraduate
Research Opportunities Program (UROP), performed an experiment, using
a factorial design, to determine the optimal drifting (exposure) time in
fresh water and optimal salinity for the critical stage 1 to stage 2 larval
molt. Hatching larvae are obtained by collecting females from the
field and maintained on laboratory water tables until hatching. Alternately,
females with maturing ovaries are brought in to the lab, maintained with
some of the males. When such females molt, the ovaries are ready
for spawing, mating occurs, and embryos are attached beneath the female
for incubation and later hatching (stage 1 larvae shown swimming at
left). Females near hatching are isolated in hatching tanks and
stage 1 larvae are collected for the experiments. In the experiments,
larvae are maintained in fresh water in treatments which vary in
the number of days in fresh water prior to introduction into seawater
of differing salinities.
We have found that a Stage 1 larva can stay (drift) in fresh water for 1-3 days and still successfully molt to Stage 2 when entering >6 ppt salinity water; molting success and survival are very low at 2 ppt. After 3 days in f.w., even at the higher salinities, the molting success declines precipitously, i.e., the larva will not molt, feed, or continue development (will not survive further).
We sampled for larvae in the Atchafalaya River both upstream at Butte La Rose (about 3 days larval drifting distance from the Atchafalaya Bay estuary) and in the Atchalaya Delta (the latter done for us by Louisiana Department of Wildlife and Fisheries, with special help from LDWF's Cassidy LeJeune). Graduate student Sara Conner analyzed the plankton samples and found only Stage-1 larvae in the river and in the Atchafalaya Delta. Abundances of larvae were significantly higher in the Delta than at Butte La Rose. Although some small amount of larval release by females occurs at Butte La Rose (about at the limit of a larva's drifting distance for successful continued development), most hatching and release of larvae occurs in the Atchafalaya Delta. This further supports our hypothesis that female M. ohione migrate down to estuaries to hatch out larvae. The results of our larval study have been published in the journal Freshwater Biology (Rome, Conner, and Bauer, 2009).


Plankton sampling for M. ohione larvae on the Atchafalaya River:
left to right,
Ray Bauer with plankton net collecting bucket
Tyler Olivier, Jim Delahoussaye, Ray Bauer
Downstream Migration of Females
Another aspect of our work is to sample reproductive-sized females throughout the year at different sites upriver in the Atchafalaya and Mississippi rivers in Louisiana to determine the distribution and abundance of females with embryos near to hatching (upriver or only near or in the coastal estuaries?). The subadult/adult population is readily sampled with baited river shrimp traps, shown below. We are helped in this sampling by the Entergy River Bend Power Plant on the Mississippi and in the Atchafalaya Delta and Mississippi (Pass-a-Loutre) estuaries by the Louisiana Dept. Wildlife and Fisheries. In Bauer and Delahoussaye (2008), we report that the relative abundance of females with embryos near hatching is far higher near or in the Atchafalaya Bay estuary than at Butte La Rose, 150 km upstream, indicating a movement (hatching migration) of reproductive females to the estuary. In the current Sea Grant project, we are extending these observations to the Mississippi River populations.


Jim Delahoussaye, Louisiana naturalist and
collaborator in the
Trap with good catch of M. ohione
project with river shrimp trap (funnel oriented downstream)
Upstream Migration of Juveniles After Larval Development
If larval development takes place
in estuaries or the open sea, the resulting small juveniles need to make
a migration back upriver to the adult f.w. habitat. Juvenile migrations,
in the form of long lines of juveniles crawling or swimming upriver along
the shore, have been more and more studied in the last few decades (e.g..,
Alan Covich and colleagues, Jeff Holmquist in Puerto Rico; various investigators
in Japan). We made a rough calculation on when such a migration might
occur based on spawing season (April-July), estimates of larval development
time from other Macrobrachium spp., distance from the Atchafalaya
Delta etc. We started looking for juveniles in midsummer; we have
now seen the very spectacular migration for the last 3 years, beginning
in mid to late July and terminating in September. Juveniles
swim right along the bank, only at night, sometimes in very high densities.
We are now studying the detailed characteristics of this migration: marine
origin of the juveniles using stable istope ratios (with Brian Fry at LSU),
growth during migration, and swimming speeds. We are studying swimming
speeds (Tyler Olivier, ULL doctoral student, with Ray Bauer and Brad Moon)
to determine how fast and how far juveniles swim. With this information,
we hope to test hypotheses about the present and past distribution of this
species and to suggest strategies for the conservation and restoration
of this species, in which human impacts on the juvenile migration are a
probable cause of the population collapse in most parts of its former range.


Juvenile shrimps (1-2 cm long) oriented and
swimming upstream along the bank
Click here to see
juveniles migrating
upriver at night
during the summer
"River Shrimp Crew"

Left to Right: Doctoral students Sara Conner and Dr. Jodi Caskey (just
graduated with a dissertation on chemical communication in shrimps began
during a previous Louisiana Sea Grant project), Ray Bauer, undergraduate
Nick Rome (UROP recipient), and doctoral student Tyler Olivier
Migration SymposiumA symposium "Life History Migrations of Freshwater Shrimps: Ecological and Adaptive Significance" was organized by Ray Bauer and Hiroshi Suzuki (Kagoshima University) at the The Crustacean Society meetings in Tokyo, Japan, Sept. 20-24, 2009. At the symposium, various speakers presented ongoing work on amphidromous shrimps from around the world and interchanged ideas about the selective forces acting on the migratory life histories of these shrimps. It was concluded that conservation of these species depends on an understanding of their migration biology. Symposium papers will be published in a proceedings volume to be published by Brill as part of its Crustacean Monograph series:

Left to Right: Ray Bauer, University of Louisiana;
Ingo Wehrtmann (back), Universidad de Costa Rica; Kazuo Mashiko (front),
Teikyo University, Japan; Klaus Anger, Biologische Anstalt Helgoland, Germany;
Marcia Synder, University of Georgia,
Athens; Yoshihisa Fujita, University of the Ryukyus, Japan; Hiroshi Suzuki,
Kagoshima University, Japan